BioDIP Publications
Proc. Natl. Acad. Sci. U.S.A. 2010 Oct 12;107(41):17633-8
Galectin-9 trafficking regulates apical-basal polarity in Madin-Darby canine kidney epithelial cells.
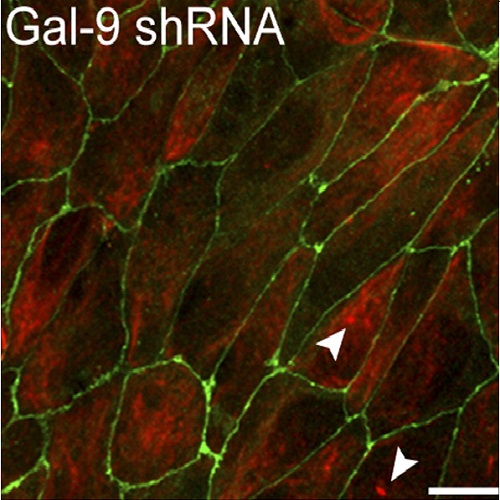
Galectins are unconventionally secreted lectins that participate in the formation of glycoprotein lattices that perform a variety of cell surface functions. Galectins also bind glycosphingolipid headgroups with as yet unclear implications for cellular physiology. We report a specific interaction between galectin-9 and the Forssman glycosphingolipid (FGL) that is important for polarizing Madin-Darby canine kidney epithelial cells. Galectin-9 knockdown leads to a severe loss of epithelial polarity that can be rescued by addition of the recombinant protein. The FGL glycan is identified as the surface receptor that cycles galectin-9 to the Golgi apparatus from which the protein is recycled back to the apical surface. Together our results suggest a model wherein such glycosphingolipid-galectin couples form a circuit between the Golgi apparatus and the cell surface that in an epithelial context facilitates the apical sorting of proteins and lipids.
Authors
Mishra R, Grzybek M, Niki T, Hirashima M, Simons K
Year
2010
PubMed ID
20861448