BioDIP Publications
Nat. Cell Biol. 2011 Sep 21;13(9):1132-8
aPKC phosphorylates NuMA-related LIN-5 to position the mitotic spindle during asymmetric division.
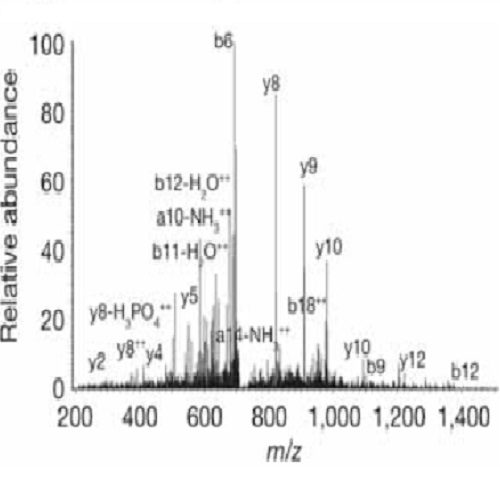
The position of the mitotic spindle controls the plane of cell cleavage and determines whether polarized cells divide symmetrically or asymmetrically. In animals, an evolutionarily conserved pathway of LIN-5 (homologues: Mud and NuMA), GPR-1/2 (homologues: Pins, LGN, AGS-3) and G? mediates spindle positioning, and acts downstream of the conserved PAR-3-PAR-6-aPKC polarity complex. However, molecular interactions between polarity proteins and LIN-5-GPR-G? remain to be identified. Here we describe a quantitative mass spectrometry approach for in vivo identification of protein kinase substrates. Applying this strategy to Caenorhabditis elegans embryos, we found that depletion of the polarity kinase PKC-3 results in markedly decreased levels of phosphorylation of a cluster of four LIN-5 serine residues. These residues are directly phosphorylated by PKC-3 in vitro. Phospho-LIN-5 co-localizes with PKC-3 at the anterior cell cortex and temporally coincides with a switch from anterior- to posterior-directed spindle movements in the one-cell embryo. LIN-5 mutations that prevent phosphorylation increase the extent of anterior-directed spindle movements, whereas phosphomimetic mutations decrease spindle migration. Our results indicate that anterior-located PKC-3 inhibits cortical microtubule pulling forces through direct phosphorylation of LIN-5. This molecular interaction between polarity and spindle-positioning proteins may be used broadly in cell cleavage plane determination.
Authors
Galli M, Muñoz J, Portegijs V, Boxem M, Grill SW, Heck AJ, van den Heuvel S
Year
2011
PubMed ID
21857670
PubMed Link
Enabled by
LMF CMCB